
Contract Manufacturing Organization (CMO), more recently referred to as a contract development and manufacturing organization (CDMO) to avoid the acronym confusion of Chief medical officer or Clinical monitoring organizations in the pharma industry, is a company that serves other companies in the pharmaceutical industry on a contract basis to provide comprehensive services from drug development through drug manufacturing. This allows the major companies to outsource those aspects of the business, which can help with scalability or can allow the major company to focus on drug discovery and drug marketing instead. CDMOs are contract manufacturers, yet they provide development as a standard part of their services. Their customers are not only expecting competitive pricing but also regulatory compliance, flexibility on the production capability, and on-time delivery.
The pharmaceutical market uses outsourcing services from providers in the form of contract research manufacturing organizations (CROs) who work on very early-stage drug development on a microscopic scale providing medicinal chemistry services. These are often called CDROs as they provide some small scale-up and later stages of drug development often preparing material ranging from hundreds of grams to multi-kilo amounts. As the drug moves through the various clinical stages, the volume tends to grow as well. Commercial scale amounts could range from metric tons. The concept of a comprehensive single-source provider from drug development through commercial manufacture of drug substances and drug products has been tried to varying success. The best-positioned service providers focus on a specific technology or dosage form and promote end-to-end continuity and efficiency for their outsourcing clients.
Global Biopharmaceutical CMO and CRO Market – Competitive Landscape
On December 7, 2023, AbbVie acquired Cerevel. On October 15, 2023, Syngene International invested about USD 100 million annually in research and biologics. On August 26, 2021, Samsung Biologics eyes up to USD 430 million in maiden bond issue. On October 28, 2020, Syneos Health to acquired Synteract a top CRO provider to emerging Biopharma. On August 5, 2020, Catalyst Clinical Research announced a merger with Ce3 Inc. to form a market-leading next-generation oncology CRO.
Some of the Key Players in the Global Biopharmaceutical CMO and CRO Market Include –
Global Biopharmaceutical CMO and CRO Market – Growth Drivers
Growing global investments in biopharmaceutical research and development serve as a primary growth driver, fostering demand for CMO and CRO services to support drug discovery and development processes. The expanding market for biologics and biosimilars fuels the demand for specialized manufacturing and research services, providing significant growth opportunities for CMOs engaged in biopharmaceutical production. Biopharmaceutical companies outsource to CMOs and CROs for efficiency gains and cost-effectiveness, allowing them to leverage external expertise, infrastructure, and flexibility in resource management. Advances in bioprocessing technologies enhance the efficiency of biopharmaceutical manufacturing, driving demand for CMOs equipped with state-of-the-art facilities and expertise in implementing innovative production methods. Collaboration partnerships between biopharmaceutical companies and service providers create growth opportunities, enabling shared resources, expertise, and risk in the dynamic landscape of drug development. The rising complexity of clinical trials especially in areas like gene and cell therapies, boosts the demand for specialized CRO services, driving growth in the factor. The growing emphasis on precision medicine and targeted therapies necessitates specialized expertise, positioning CROs as key contributors in conducting focused clinical trials and research activities.
Global Biopharmaceutical CMO and CRO Market – Restraints
The biopharmaceutical CMO and CRO market faces challenges associated with stringent and evolving regulatory requirements, which can increase operational complexities and compliance costs. Intellectual property issues pose a restraint, as collaboration with CMOs and CROs involves sharing proprietary information, leading to concern about data protection and potential disputes over ownership rights. Setting up and maintaining state-of-the-art biopharmaceutical manufacturing and research facilities requires significant initial capital investment, which can be a restraint for both existing and new entrants in the CMO and CRO market. CMOs are often highly dependent on the pipeline of their client biopharmaceutical companies. Fluctuations or delays in client projects can impact the revenue streams and overall sustainability of CMOs. The industry is susceptible to global supply chain disruptions, as seen with events like the COVID-19 pandemic, affecting the timely delivery of material and impacting the efficiency of CMO and CRO operations. CROs face the risk of clinical trial failures, which can occur due to factors beyond their control, such as unforeseen safety issues or lack of efficiency, leading to financial setbacks and damage to reputation. Intense competition among CMOs and CROs can result in pricing pressures impacting profit margins. Maintaining competitive pricing while delivering high-quality services is a constant challenge in the market. The industry faces challenges in attracting and retaining skilled professionals, leading to talent shortages and turnover. This can impact the consistency and quality of services provided by CMOs and CROs.
Global Biopharmaceutical CMO and CRO Market – Opportunities
The increased global investments in biopharmaceutical research and development present substantial opportunities for CMOs and CROs to offer specialized services, driving innovation and drug development. Opportunities arise with the increasing adoption of advanced technologies, such as artificial intelligence, data analytics, and automation, enhancing the efficiency and productivity of CMO and CRO services. Biopharmaceutical companies increasingly outsource early-stage development activities to CROs, providing an opportunity for these service providers to engage in the critical phases of drug discovery and development.
Global Biopharmaceutical CMO and CRO Market – Geographical Insight
The global biopharmaceutical CMO and CRO market is segmented into regions such as North America, Latin America, Europe, Asia-Pacific, the Middle East & Africa. North America is the largest market for biopharmaceutical CMOs and CROs due to key players and a robust infrastructure supporting biopharmaceutical development. Europe is witnessing a growing presence in the biopharmaceutical outsourcing market with increased investments in research and development activities. The Asia-Pacific region is emerging as a significant hub for biopharmaceutical outsourcing, fueled by a combination of skilled workforce, cost advantages, and advancing regulatory framework.
Global Biopharmaceutical CMO and CRO Market – Key Development
Research Methodology: Aspects
Market research is a crucial tool for organizations aiming to navigate the dynamic landscape of customer preferences, business trends, and competitive landscapes. At Cognizance Market Research, acknowledging the importance of robust research methodologies is vital to delivering actionable insights to our clientele. The significance of such methodologies lies in their capability to offer clarity in complexity, guiding strategic management with realistic evidence rather than speculation. Our clientele seek insights that excel superficial observations, reaching deep into the details of consumer behaviours, market dynamics, and evolving opportunities. These insights serve as the basis upon which businesses craft tailored approaches, optimize product offerings, and gain a competitive edge in an ever-growing marketplace.
The frequency of information updates is a cornerstone of our commitment to providing timely, relevant, and accurate insights. Cognizance Market Research adheres to a rigorous schedule of data collection, analysis, and distribution to ensure that our reports reflect the most current market realities. This proactive approach enables our clients to stay ahead of the curve, capitalize on emerging trends, and mitigate risks associated with outdated information.
Our research process is characterized by meticulous attention to detail and methodological rigor. It begins with a comprehensive understanding of client objectives, industry dynamics, and research scope. Leveraging a combination of primary and secondary research methodologies, we gather data from diverse sources including surveys, interviews, industry reports, and proprietary databases. Rigorous data analysis techniques are then employed to derive meaningful insights, identify patterns, and uncover actionable recommendations. Throughout the process, we remain vigilant in upholding the highest standards of data integrity, ensuring that our findings are robust, reliable, and actionable.
Key phases involved in in our research process are mentioned below:

Understanding Clients’ Objectives:
Extensive Discussions and Consultations:
Industry and Market Segment Analysis:
Target Audience Understanding:
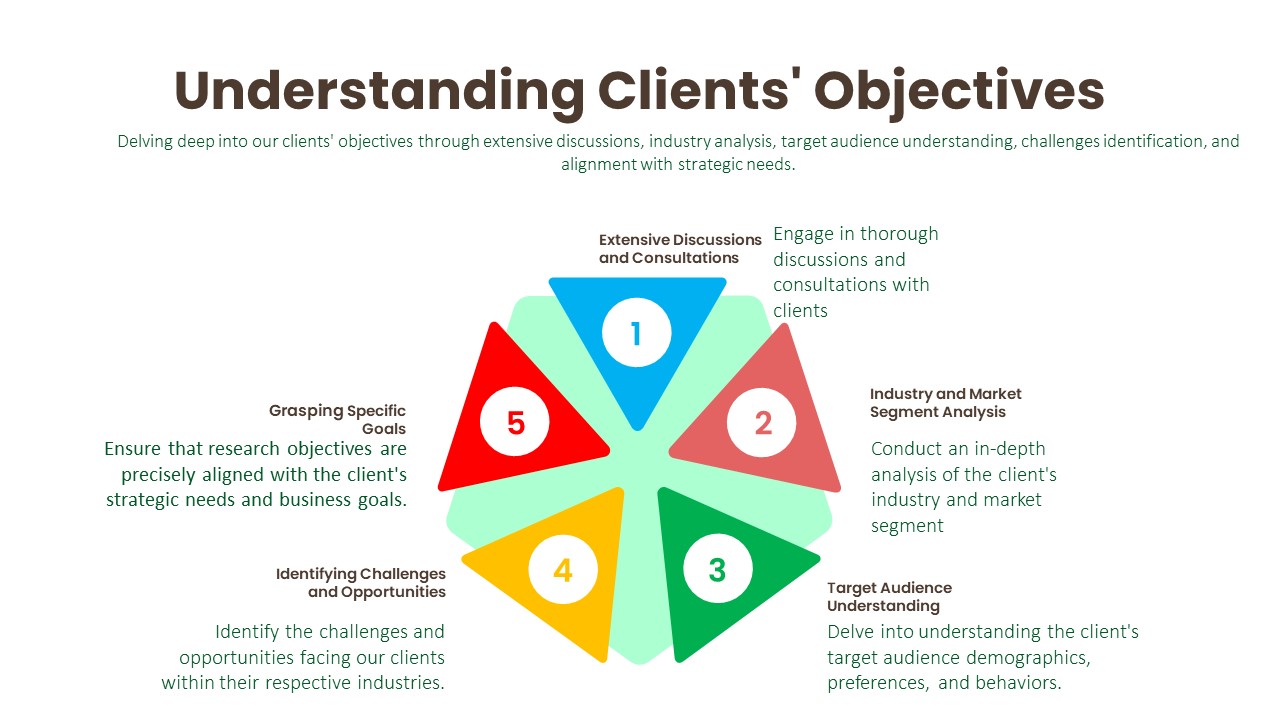
Identifying Challenges and Opportunities:
Grasping Specific Goals:
Data Collection:
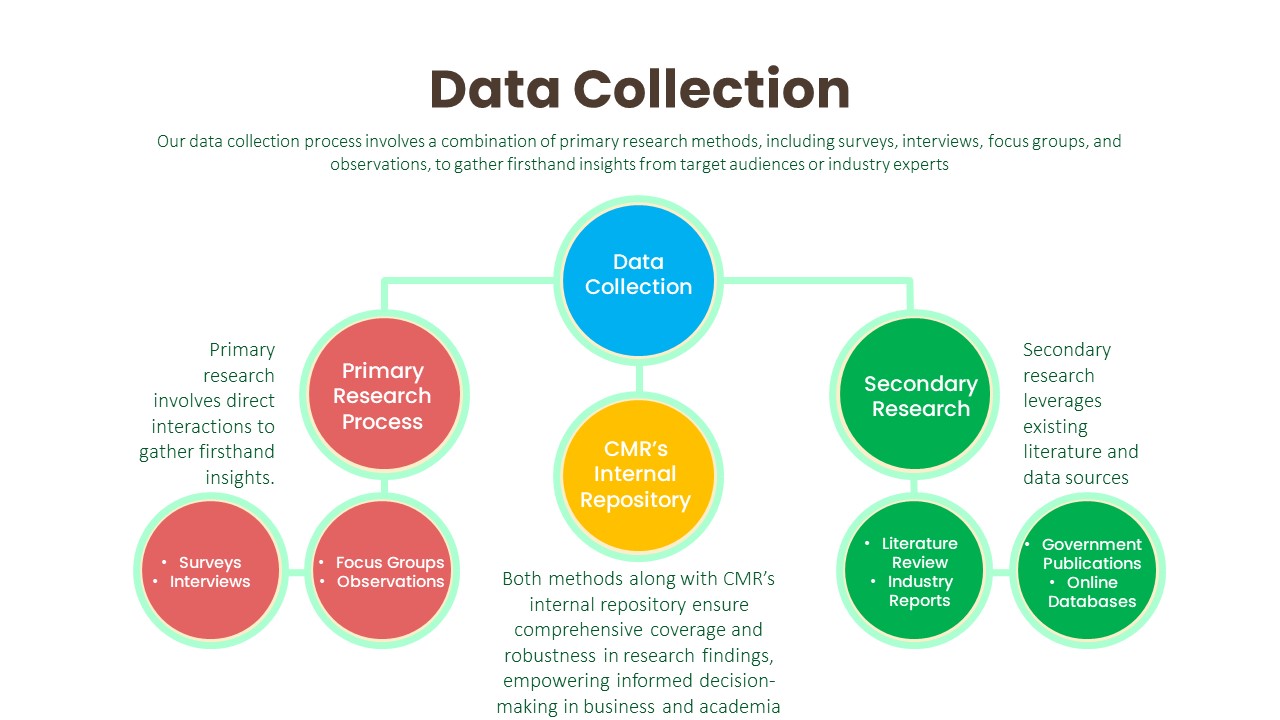
Primary Research Process:
Secondary Research Process:


Data Analysis:
The data analysis phase serves as a critical juncture where raw data is transformed into actionable insights that inform strategic decision-making. Through the utilization of analytical methods such as statistical analysis and qualitative techniques like thematic coding, we uncover patterns, correlations, and trends within the data. By ensuring the integrity and validity of our findings, we strive to provide clients with accurate and reliable insights that accurately reflect the realities of the market landscape.

Transformation of Raw Data:
Utilization of Analytical Methods:
Statistical Analysis:
Qualitative Analysis Techniques:
Integrity and Validity Maintenance:
Data Validation:
The final phase of our research methodology is data validation, which is essential for ensuring the reliability and credibility of our findings. Validation involves scrutinizing the collected data to identify any inconsistencies, errors, or biases that may have crept in during the research process. We employ various validation techniques, including cross-referencing data from multiple sources, conducting validity checks on survey instruments, and seeking feedback from independent experts or peer reviewers. Additionally, we leverage internal quality assurance protocols to verify the accuracy and integrity of our analysis. By subjecting our findings to rigorous validation procedures, we instill confidence in our clients that the insights they receive are robust, reliable, and trustworthy.
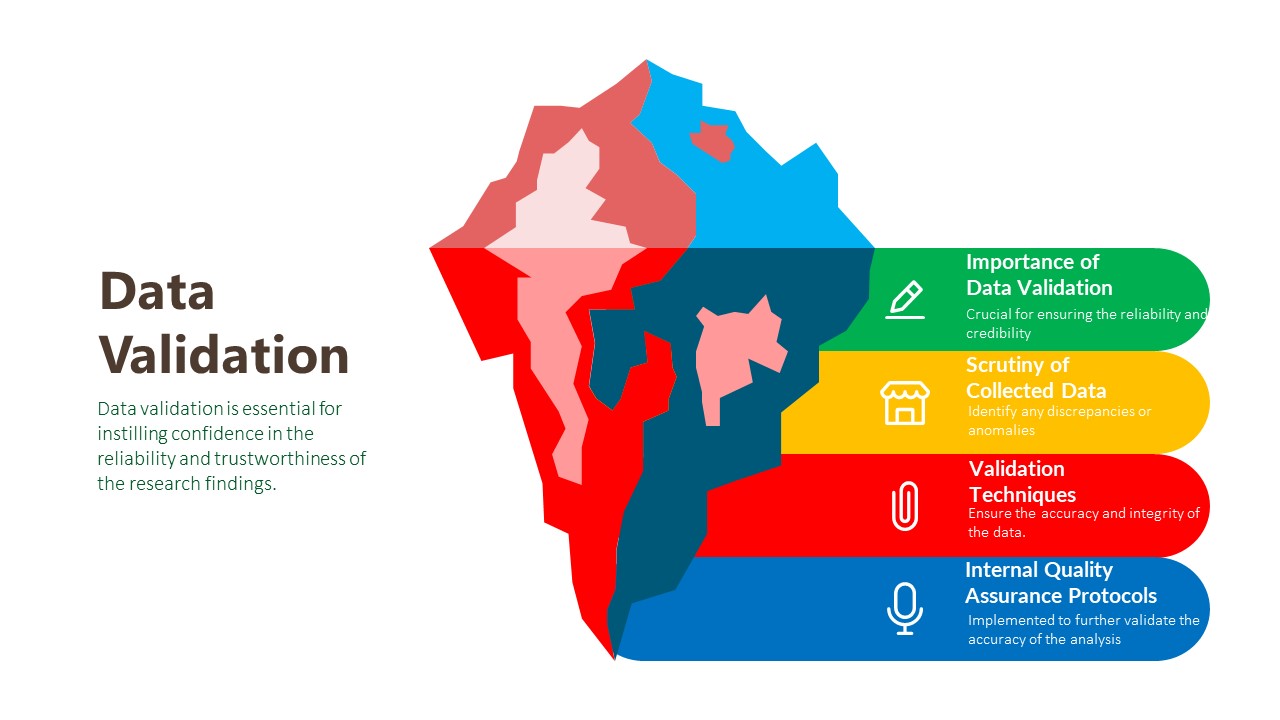
Importance of Data Validation:
Scrutiny of Collected Data:
Validation Techniques:
Internal Quality Assurance Protocols:
We can customize every report – free of charge – including purchasing stand-alone sections or country-level reports
We help clients to procure the report or sections of the report at their budgeted price. Kindly click on the below to avail

Cognizance market research is continuously guiding customers around the globe towards strategies for transformational growth. Today, businesses have to innovate more than ever before, not just to survive, but to succeed in the future

© 2023 All rights Reserved. Cognizance Market Research